 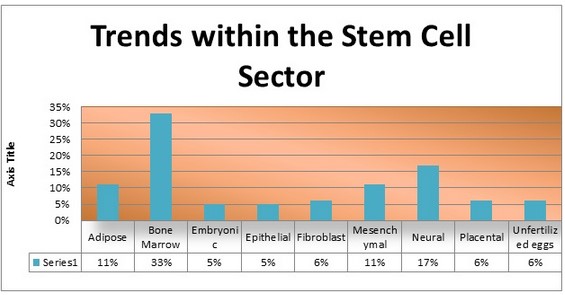
The NurOwn clinical program has generated valuable insights into the pathology of ALS, as well as disease progression and treatment. Three CSF biomarkers were predictive of clinical outcomes in NurOwn-treated participants– neurofilament light (NfL), galectin-1, latency associated peptide of TGF-beta1 (LAP or TGF-b). Biomarker patterns were consistent across all NurOwn-participants, including in those with Advanced ALS disease where clinical scales, such as the ALS Functional Rating Scale, have demonstrated measurement challenges. 
Biomarker data in all trial participants showed consistent biological patterns of NurOwn reducing markers of inflammation and neurodegeneration, and increasing neuroprotective markers relative to placebo. NurOwn's clinical program also included the largest cerebrospinal fluid (CSF) biomarker study ever done in ALS, strong and consistent biomarker data, which are predictive of clinical response in the trial, span pathways that are important to ALS (neuroinflammation, neurodegeneration, neuroprotection), and align with NurOwn's mechanism of action. 
Additionally, a post-hoc sensitivity analysis of patients across threshold of >26 through ≥35 on the ALSFRS-R highlighted that NurOwn-treated patients retain, on average, two points of function more compared to placebo - clinically meaningful preservation and important for quality of life for a person living with ALS and their loved ones. With a statistically significant difference on a key endpoint (change from baseline in ALSFRS-R). In a pre-specified group of participants with an ALSFRS-R score ☳5, there was larger treatment effect across all endpoints with NurOwn compared to placebo, which aligned with historical trials and the study power assumptions. A thorough analysis of NurOwn Phase 3 data shows evidence of clinically meaningful effectiveness in ALS participants who have not progressed to advanced levels of disease progression. The Phase 3 pivotal trial NurOwn did not reach statistical significance on the primary or secondary endpoints, likely due to a "floor effect," which confounds measurement of disease progression in patients with more advanced disease. BrainStorm's long-term commitment to ALS is demonstrated in preclinical research and a series of clinical studies, all of which have been published in peer-reviewed journals. 
The lead program for NurOwn is for the treatment of ALS, which is under FDA review. MSC-NTF cells are harvested from each person with ALS and are manufactured using an innovative and proprietary process to secrete neurotrophic factors to target specific neurodegenerative diseases. The NurOwn® technology platform (autologous MSC-NTF cells) represents a promising investigational therapeutic approach to targeting disease pathways important in neurodegenerative disorders. We believe this approach honors the needs of those living with ALS and offers the greatest promise for BrainStorm to fulfill our commitment to the ALS community." As is the case with most ALS research, our clinical program generated complex results, which deserve a thoughtful and holistic review by scientists, ALS experts, FDA reviewers, advocates, and patients. We remain confident in NurOwn and believe our data support regulatory approval. Stacy Lindborg, Ph.D., BrainStorm co-CEO, commented: "We welcome the opportunity to present our data at the forthcoming ADCOM. 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
